 Then calculate the number of valence electrons used in this drawing. Given: molecular formula and molecular geometryĪ Draw a structure for benzene illustrating the bonded atoms. Use resonance structures to describe the bonding in benzene. For carbon bonded to a more electronegative non-metal X, such as nitrogen, oxygen, sulfur or the halogens, each C-X. This means that every C-H bond will decrease the oxidation state of carbon by 1. The benzene molecule (C 6H 6) consists of a regular hexagon of carbon atoms, each of which is also bonded to a hydrogen atom. To calculate the oxidation state for carbon, use the following guidelines: In a C-H bond, the H is treated as if it has an oxidation state of +1. (Note: N is the central atom.)īenzene is a common organic solvent that was previously used in gasoline it is no longer used for this purpose, however, because it is now known to be a carcinogen. Draw three Lewis electron structures for CNO − and use formal charges to predict which is more stable. Benzene is a six-carbon aromatic annulene in which each carbon atom donates one of its two 2p electrons into a delocalised pi system. Salts containing the fulminate ion (CNO −) are used in explosive detonators. 8.2 Illustrated are four ionsA, B, X, and Yshowing their relative ionic radii. 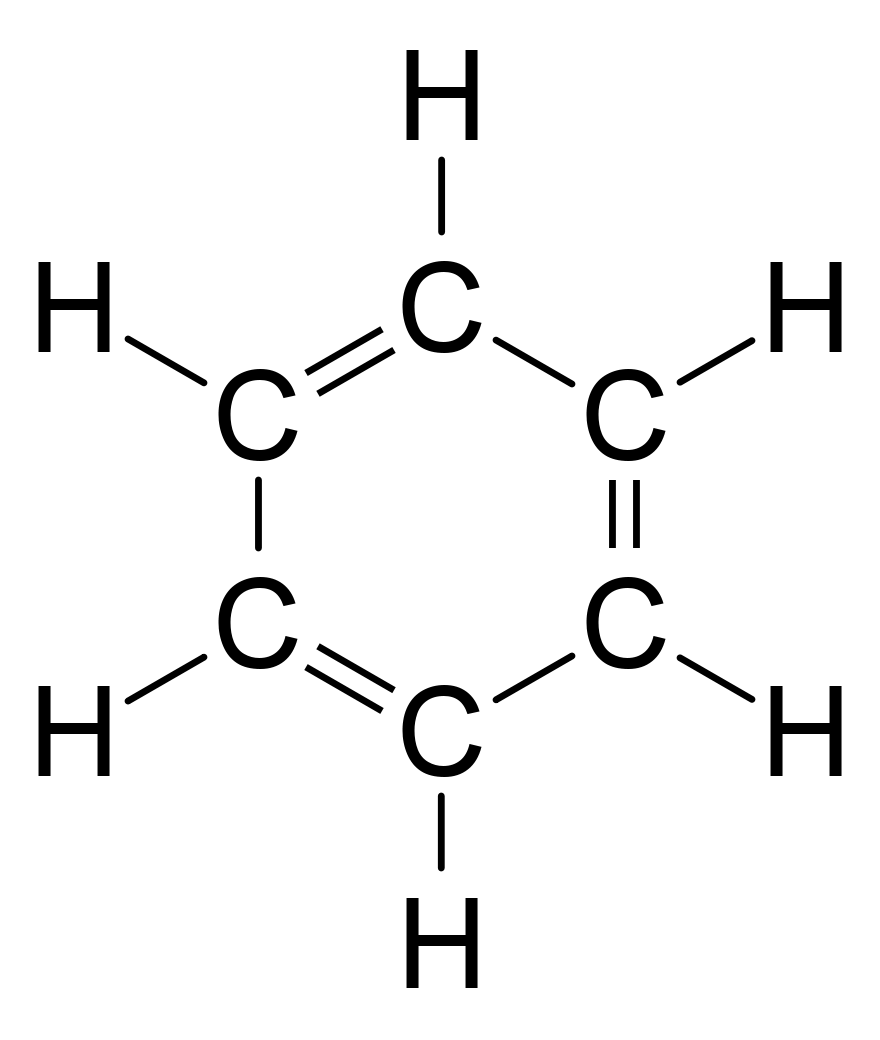 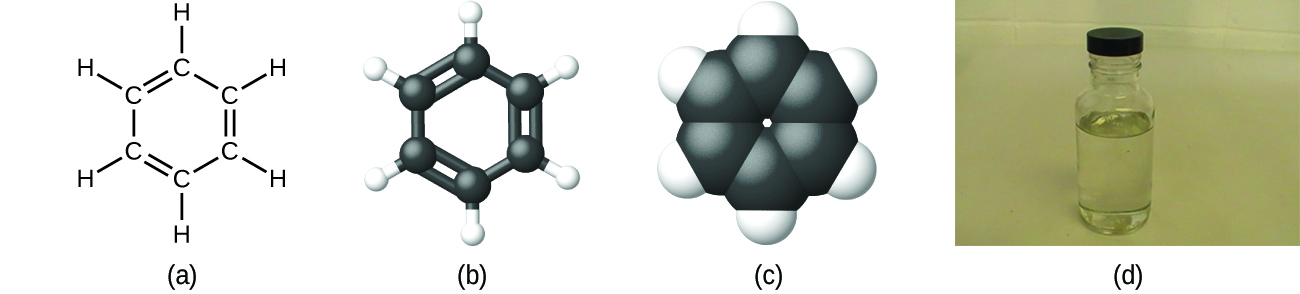
8.1 For each of these Lewis symbols, indicate the group in the periodic table in which the element X belongs: Section 8.1. \right )=-1 \) In (c), nitrogen has a formal charge of −2.Ĭ Which structure is preferred? Structure (b) is preferred because the negative charge is on the more electronegative atom (N), and it has lower formal charges on each atom as compared to structure (c): 0, −1 versus +1, −2. Rules for writing the Lewis structure of a molecule: 1) MF, constitution, and net charge must be known 2) Draw the dot structures of all the atoms of the MF. CHEMISTRY THE CENTRAL SCIENCE 8 BASIC CONCEPTS OF CHEMICAL BONDING EXERCISES. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
